Teachers' Domain - Digital Media for the Classroom and Professional Development
User: Preview
Source: Thomas J. Greenbowe and the Iowa State University Chemical Educational Group.
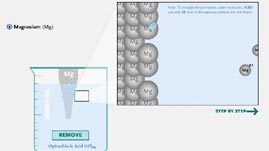
In this interactive activity adapted from Iowa State University, observe the chemical reaction that occurs when different metals are dipped in a solution of hydrochloric acid. Hydrochloric acid dissolves some metals to form oxidized metal chlorides and hydrogen gas, the latter of which is visible in the animation as bubbles floating to the surface. Depending on their atomic structure and how readily they give up electrons when exposed to hydrogen ions in an acidic solution, certain metals, including zinc and magnesium, corrode more easily than others.
An acid is a chemical compound that, when dissolved in water, has a pH of less than 7.0. Acidic compounds contain positively charged hydrogen ions—hydrogen atoms containing protons but no electrons—which readily react with other compounds. Examples of common acids are acetic acid, or vinegar, and sulfuric acid, which is used in car batteries. Monoprotic acids, which include hydrochloric acid, only give up one proton per acid molecule, while polyprotic acids can give up more than one proton per acid molecule.
When bare-metal surfaces are immersed in an acidic aqueous solution (a solution in which the solvent is water), the hydrogen ions in the acid attract and bond with negatively charged electrons from the metal to produce what is called an oxidation-reduction reaction. Hydrogen gas, which forms as two released electrons attach to two hydrogen ions, bubbles off, and metal atoms (now positively charged ions) are released into the solution. Through this oxidation-reduction reaction, the metal is said to corrode, or gradually wear away.
Metals consist of stacked layers of tightly packed, interacting atoms arranged in geometric patterns. Whereas non-metals tend to gain electrons in chemical reactions, metals tend to lose them. The gauge of just how readily a metal loses electrons is known as its redox potential, which is determined by its atomic structure.
Electrons inhabit various energy levels, or shells. The electron configuration shown in the periodic table indicates how many electrons are found in each shell, from innermost to outermost. Atoms of elements in the left-hand column have one electron in their outer shell, while atoms of elements in the right-hand column have eight electrons in their outer shell. Generally speaking, single electrons in an outer shell can easily be taken away from the atom with the application of very little energy. This makes atoms of elements in the left-hand column very reactive. On the other hand, it is very difficult to add or remove electrons from an atom that has eight electrons in its outer shell. The atoms of these elements, found in the column to the far right, are non-reactive. Knowing this helps explain why some metals, such as tin and copper, corrode less readily than others, such as magnesium and iron.
Because some metals are more prone to corrosion, engineers have learned to use certain techniques during an item's manufacture to protect it from structural decay. One method involves plating, or painting over a metal with a coating. This technique prevents electrochemical reactions from occurring. When exposed to oxygen in air or water, for example, two elements, aluminum and chromium, form a self-renewing, microscopically thin layer, known as an oxide film, which protects the underlying metal from corrosion. The alloy stainless steel, known for its sleek, shiny surface and tremendous strength, contains a high concentration of chromium.
 Loading Standards
Loading Standards Teachers' Domain is proud to be a Pathways portal to the National Science Digital Library.
Teachers' Domain is proud to be a Pathways portal to the National Science Digital Library.
