Teachers' Domain - Digital Media for the Classroom and Professional Development
User: Preview
Source: The ASPIRE Lab, University of Utah
In this interactive activity adapted from the University of Utah’s ASPIRE Lab, learn about wavelength. Use a ruler to measure the distance from crest to crest on a simple waveform to find its wavelength. Then, investigate how wavelength varies across the electromagnetic spectrum and how the colors of visible light are related to wavelength.
Within the spectrum of visible light (all the light that people can see), there is a range of colors of light. However, visible light is just one small portion of the entire electromagnetic spectrum, which includes an even wider range of different types of light, including radio waves, microwaves, infrared light, ultraviolet light, X-rays, and gamma rays. But what is it that makes these various kinds of light different from one another?
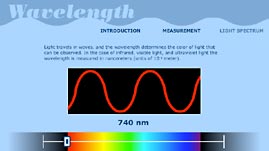
To answer this, first we must understand that all forms of light are electromagnetic waves. Scientists have agreed to visually represent all waves as having a sinusoidal shape—the familiar S-shape. The points of maximum displacement in the positive or negative direction are known as the "crests" and "troughs" of a wave.
The length of one complete wave cycle—for example, from crest to crest—is called the wavelength. Different types of light have different wavelengths. For example, visible light falls within the range of wavelengths from about 400 nanometers to 700 nanometers (from violet to red light). Each category of light in the electromagnetic spectrum is simply a band of wavelengths.
One characteristic of waves is frequency, the measure of the rate of oscillation of a wave. The frequency is a measure of the number of wave cycles that occur in a given amount of time and is related to the wavelength; the product of wavelength and frequency equals the speed of a wave.
As a result, because all light travels at the same speed, there is a clear relationship between the wavelength and frequency of light—the shorter the wavelength of light, the higher its frequency. The electromagnetic spectrum is the range of all possible wavelengths and frequencies, spanning from very long wavelengths (low frequencies) to very short wavelengths (high frequencies). For example, radio waves have long wavelengths (up to thousands of kilometers long) and X-rays have short wavelengths (less than one-billionth of a meter). By convention, we usually describe radio waves in terms of frequency; however, optical and infrared light are usually described in terms of wavelength, and X-rays and gamma rays are described in terms of energy.
 Loading Standards
Loading Standards Teachers' Domain is proud to be a Pathways portal to the National Science Digital Library.
Teachers' Domain is proud to be a Pathways portal to the National Science Digital Library.
