Teachers' Domain - Digital Media for the Classroom and Professional Development
User: Preview
Source: Wisconsin Online Resource Center
In this interactive activity adapted from the Wisconsin Online Resource Center, explore methods of heat transfer and classify examples from everyday life. Click on each of the three methods of heat transfer (conduction, convection, and radiation) to see a description and animated illustration of that method.
Heat is energy that is transferred between two objects or substances of different temperatures; heat typically flows from a warmer material to a cooler material. Generally, when heat is transferred to a material, the motion of its particles speeds up and its temperature increases.
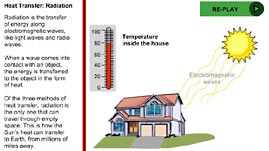
There are three methods of heat transfer: radiation, conduction, and convection. Radiation transfers energy by electromagnetic waves, a method in which heat can transfer even in the absence of matter (through outer space, for example). When electromagnetic radiation strikes an object, the energy carried by the electromagnetic wave is transferred to the object, causing the particle motion within the object to increase. For example, a microwave oven emits microwave radiation to transfer heat to food. Similarly, the reason that you can feel the warmth of an object at a distance, such as from the Sun or a light bulb, is due to the transfer of heat by radiation. While all matter emits and absorbs electromagnetic radiation, some materials are better at absorbing radiation than others; shiny surfaces, for example, tend to reflect rather than absorb radiation.
Conduction transfers heat through direct contact. If two objects are placed in contact with each other, heat flows from the warmer object (with faster-moving particles) to the cooler object (with slower-moving particles) by the direct interaction of the particles. When the faster particles collide with the slower particles, they transfer some of their energy to the slower particles. For example, when a hot pan is placed on a counter, the counter increases in temperature as the faster-moving molecules of the pan collide with and increase the motion of the molecules of the counter. At the same time, the molecules of the pan slow down, and the temperature of the pan drops. Some materials, such as metals, are good conductors of heat while other materials, such as glass, wood, plastic, and air, are not. Materials that are not good at transferring heat by conduction are known as insulators.
Convection transfers heat through the movement of fluids or gases in circulation cells. A pot of water heated on a stove provides an example. The pot itself, and then water at the bottom, becomes heated by conduction. When water is heated, it expands, becomes less dense, and rises up through the surrounding cooler water. The cooler, denser water then sinks to the bottom of the pot where it, in turn, is heated. The convection current—the circulating path of hot water rising and cold water sinking—transfers heat by actually moving the warmer water to a new area. It also forces the hot water to mix with the cooler water and increases conduction by bringing the cool water to the bottom of the pot.
 Loading Standards
Loading Standards Teachers' Domain is proud to be a Pathways portal to the National Science Digital Library.
Teachers' Domain is proud to be a Pathways portal to the National Science Digital Library.
